Int J Chem Res, Vol 10, Issue 2, 12-17Research Article
FE(ACAC)₃-CATALYZED ONE-POT SYNTHESIS OF SUBSTITUTED QUINOXALINE DERIVATIVES
SUNIL S. CHOUDHARE1 , SANTOSH PADGHAN1, SURAJ ADE2, MANOJKUMAR CHOPADE1*
, SANTOSH PADGHAN1, SURAJ ADE2, MANOJKUMAR CHOPADE1*
1Department of Chemistry, Sant Dnyaneshwar Mahavidyalaya, Soegaon, Chhatrapati Sambhaji Nagar-431120, Maharashtra India. 2Department of Chemistry, Maulana Azad College, Chhatrapati Sambhaji Nagar, Maharashtra India
*Corresponding author: Manojkumar Chopade; *Email: [email protected]
Received: 23 Oct 2025 Revised and Accepted: 09 Mar 2025
ABSTRACT
Objective: The study was to develop an efficient and practical method for the synthesis of substituted quinoxaline derivatives, which are known for their significant biological activities and structural versatility, and serve as important building blocks in organic synthesis.
Methods: A series of substituted quinoxaline derivatives were synthesized through a one-pot cyclocondensation reaction between substituted phenyl diamines and phenacyl bromide. The reaction was catalyzed by Fe(acac)₃ and carried out at 60 °C for a duration of 50–80 min.
Resuts: The reaction successfully afforded quinoxaline derivatives (3a–3m) in good to excellent yields ranging from 88–94%. The synthesized compounds exhibited high purity, demonstrating the efficiency of the developed protocol.
Conclusion: The present methodology offers several practical advantages, including a simple one-pot procedure, facile isolation of products, high yields, and an efficient cascading cyclocondensation process. This approach provides an effective and reliable route for the synthesis of substituted quinoxaline derivatives.
Keywords: Fe(acac)3, Phenyldiamine, Cyclocondensation, Phenacyl bromide, Quinoxaline
© 2026 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (http://creativecommons.org/licenses/by/4.0/)
DOI: http://dx.doi.org/10.22159/ijcr.2026v10i2.322 Journal homepage: https://ijcr.info/index.php/journal
INTRODUCTION
Quinoxalines represent a significant class of nitrogen-containing heterocyclic compounds that have attracted considerable attention in recent decades due to their diverse biological and pharmaceutical potentials [1]. Although these compounds are rarely found in natural sources, they have been widely synthesized through a variety of chemical methodologies [2]. With the growing emphasis on environmental sustainability and green chemistry, significant efforts have been directed toward the development of eco-friendly and efficient synthetic strategies for quinoxaline derivatives. These approaches often utilize recyclable catalysts, non-toxic solvents, and mild reaction conditions, aligning with the principles of green chemistry [3].
In recent years, quinoxalines have emerged as highly promising scaffolds in medicinal chemistry owing to their broad spectrum of biological activities [4]. Numerous quinoxaline-based derivatives have demonstrated potent bioactivities, including antitubercular [5], anti-inflammatory, antioxidant [6], antifungal, anticancer, anti-HIV [7] and antiprotozoal properties [8]. Their structural versatility and ease of functionalization have made them valuable templates for drug design and development.
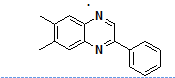
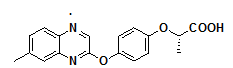
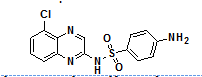
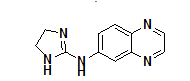
Moreover, many clinically approved drugs and investigational agents incorporate a quinoxalines moiety as a key pharmacophore unit within their molecular framework, which significantly contributes to their therapeutic efficacy. The incorporation of the quinoxaline nucleus often enhances target specificity, bioavailability and metabolic stability. Examples of such drugs are illustrated in fig. 1, showcasing the relevance of this heterocyclic core in modern pharmaceutical chemistry.
AG1295 tyrosine kinase |
XK-469 anticancer |
Chloroquinoxaline sulfonamide Anticancer |
Quinacillin Antibiotic |
Brimonidine Antihypertensive |
Varenicline Antismoking |
Fig. 1: Quinoxaline core with natural and bioactive compounds
Quinoxalines are nitrogen-containing heterocycles found in various natural and synthetic compounds. They serve as key building blocks in heterocyclic synthesis and play a crucial role in organic and medicinal chemistry. Recently, numerous methods have been developed to diverse synthesis of quinoxaline derivatives via transition metals [9]. In recent years, focusing on several environmental issues and considering the pharmacological importance of quinoxaline scaffolds there are numerous synthetic methods have been reported, Some of them are the condensation of o-pheneylenediamine with 1,2-dicarbonyl compounds, Among the aforementioned protocols, condensation of o-pheneylenediamine with phenacyl bromides catalyzed by solid acid catalyst[10]and using transition metal catalysts as well [11] is also highly preferred which includes (NH4)6Mo7O24·4H2O [12], Na2PdP2O7 [13] and Ga(ClO4)3 [14], Synthesis of spiro-indeno[1,2-b]quinoxalines via a g-Fe2O3@Oxo-triazolidin-sultone Nano catalyst [15], CuBr/O2 catalytic system [16]. Additionally, oxidative cyclization betweendeoxybenzoins and 1,2-pheneylenediaminein the presence of a catalytic amount of a Cu(II)-complex of a zwitterioniccalix [17], Ni(II)/1,10-phenanthroline-catalyzeddehydrogenativecoupling reaction for the synthesis of quinoxalines [18] and quinoxaline synthesis using heterogeneous solid zinc oxide nanoparticles loaded on mesoporous silica (ZnO-KIT-6) [19] have also been reported.
MATERIALS AND METHODS
All chemicals and solvents were dried before use and purchased commercially and used exactly as supplied. Chemicals and solvents were procured from Sigma Aldrich and Spectrochem. Melting points were determined in open capillary and are uncorrected. Reactions were monitored by thin layer chromatography (TLC) on silica gel plates (GF 254) using UV light to visualize the course of the reactions. 1H NMR spectra and 13C NMR spectra were recorded Bruker Avance 300-400 (FT-NMR) and Bruker DRX-300 instruments, respectively, using CDCl3 and DMSO-d6 as solvent. Chemical shifts are reported in δ ppm with TMS as internal standard. High-resolution mass spectra (HRMS) were obtained using the Agilent 6520 (Q-TOF) ESI-HRMS instrument. Routine monitoring of reaction was performed by TLC using 0.25 mm E. Merck precoated silica gel TLC plates (60 F254) hexane: ethyl acetate as eluent.
General experimental procedure for synthesis of quinoxalines
In a round-bottom flask, a mixture of phenacyl bromide (2a–2m, 0.001 mol) and Fe(acac)₃ catalyst (40 mg) was dissolved in ethanol (5 ml) and stirred at room temperature for 10 min. Subsequently, o-pheneylenediamine (1a–1b), 0.001 mol) was added slowly to the reaction mixture, and the resulting homogeneous mixture was stirred at 60 °C for the stipulated time. The progress of the reaction was monitored by thin-layer chromatography (TLC). The reaction mixture was diluted with ethyl acetate (20 ml) anddiluted HCl (5 ml, 0.1 N). The combined organic layers were dried using anhydrous MgSO4, filtered, and the solvent was removed by evaporation. The crude product was purified by crystallization using ethanol to afford the pure 2-pheny quinoxalines (3a–3k). All desired product characterised by IR, H1-NMR, C13NMR and melting points of the desired products were found to be in good agreement with those reported in the literature.
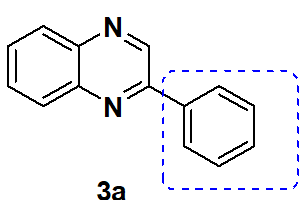
2-Phenylquinoxaline (3a) [8]
Isolated as a Yellow Solid. Yield 90 %; Mp. 78 C(Lit[1, 4, 21]75-76 C). IR (KBr, cm-1): νmax 3447, 3059, 2921, 2852, 1631, 1544, 1487, 1314, 1224, 1028, 956, 766 cm.-[11]H NMR (500 MHz, Chloroform-d) δ 9.32 (s, 1H, =CH), 8.11-8.21 (m, 4H, Ar-H), 7.71-7.80 (m, 2H), 7.50-7.59 (m, 3H, Ar-H). 13C NMR (100 MHz, CDCl3) δ (ppm): 151.9, 143.5, 142.5, 141.7, 136.9, 130.3, 130.3, 129.8, 129.6, 129.2, 127.7 HRMS (ESI)+m/z calcd. for C14H10N2 (M+H)+: 207.0923; found 207.0926.
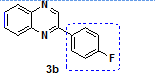
2-(4-Fluorophenyl)Quinoxaline (3b) [4]
Isolated as a Yellow Solid, Yield 90%; Mp. 113-119 C (Lit[4, 21]112-118 C). IR (KBr, cm-1): ῡmax 3421, 2927, 1633, 1583, 1534, 1475, 1418, 1101, 1073, 955, 830, 759 cm.-11H NMR (300 MHz, CDCl3) δ 9.25 (s, 1H, =CH), 8.07-8.19 (m, 4H, Ar-H), 7.68-7.78 (m, 2H, Ar-H), 7.18-7.24 (m, 2H, Ar-H). 13C NMR (75 MHz, CDCl3) δ (ppm): 165.9, 162.6, 150.7, 142.9, 142.2, 141.5, 133.0, 132.9, 130.4, 130.2, 129.6, 129.6, 129.5, 129.2, 116.4, 116.1. HRMS (ESI)+m/z calcd. for C14H9FN2 (M+H)+: 225.0823; found 225.0852
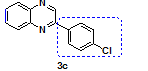
2-(4-Chloro phenyl)-Quinoxaline (3c)lit [1, 21]
Isolated as a Yellow solid, Yield 94%; Mp: 128-132 °C; IR (KBr, cm-1): νmax2924, 1591, 1537, 1479, 1310, 1122, 1043, 958, 832, 753 cm-1. 1H NMR (400 MHz, CDCl3) δ 9.28 (s, 1H, =CH), 8.09-8.15 (m, 4H), 7.72-7.80 (m, 2H), 7.51 (d, J = 8.5 Hz, 2H). 13C NMR (100 MHz, CDCl3) δ (ppm): 150.7, 142.9, 142.3, 141.8, 136.7, 135.3, 130.6, 129.9, 129.7, 129.5, 129.3, 128.9. (CH3). MS (ESI):m/z calcd for C14H9ClN2 [M+H]+: 241.3113, found 241.0512
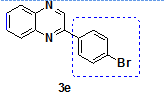
2-(4-Bromo phenyl)-Quinoxaline (3e)lit [4, 21]
Isolated as a pale yellow solid, Yield 92%; Mp. 128-134 °CIR (KBr, cm-1): νmax 3443, 2925, 1634, 1583, 1536, 1481, 1421, 1307, 1121, 1070, 954, 827, 710 cm-[11]H NMR (400MHz, CDCl3) δ 9.26 (s, 1H =CH), 8.04-8.13 (m, 4H, Ar), 7.65-7.79 (m, 4H, Ar). 13C NMR (100 MHz, CDCl3) δ (ppm): 150.7, 142.8, 142.3, 141.8, 135.7, 132.4, 130.6, 129.9, 129.7, 129.3, 129.1, 125.1. MS (ESI): m/z calcd for C14H9BrN2 [M+H]+: 287.21, found 287.07
RESULTS AND DISCUSSION
The synthesis of quinoxaline were the model reaction of o-pheneylenediamine (1a) and phenacyl bromide (2c) was carried out in presence iron acetyl acetonate (Fe(acac)3) stirred at 60 °C for 60 min, with good yield 92 % with high purity productsas shown in scheme 1.
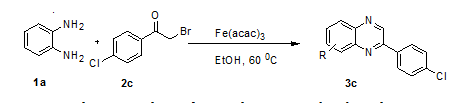
Scheme 1: Synthesis of quinoxalines using Fe(acac)3 catalyst
The optimization of the reaction was performed by varying the reaction parameters, such as reaction time, solvent, and temperature. It was observed that the quinoxaline formation in ethanol solvent proceeds with an excellent yield at 60 °C for 50 min (table 1, entry 3).
The IR spectrum of compound quinaxoline indicates presence of two bands at 3447 and 3059 cm–1. (CAr), 1631, 1544, (C=N), which corresponds to Imine and Aromatic region respectively. 2-Phenyl quinoxalines 3a characterized by 1H-NMR spectrum, the Aromatic peak-appears as a doublet peak at δ 8.21 – 8.11 due to Ar-N and Ar-H of ten proton and The very important signal is obtained at 9.32 δ, which is singlet of Aromatic ring HC=N. The 2-Phenyl quinoxalines 3a compound charecretised by 13C NMR 151.9(C=N), (CAr), 143.5 (C, S-C=N), 142.5 indicates that formation of Quinoline ringlit [1-4, 21].
First, we optimized the solvent for synthesizing the target molecule. The model reaction initially water was used as the solventyielded low product. To improve the yield of this, we Screen different solvents, including protic (water, methanol, ethanol) solvents and aprotic (dioxane, Chloroform, DCM, acetonitrile). The results are summarized in table 1. The highest yield was obtained with ethanol (table 1, entry 3), while protic solvents resulted in higher yields than aprotic ones. Among them, ethanol proved most effective for Quinoxalinesynthesis, making it the optimal solvent table 1.
Table 1: Screening of reaction condition with respect to solvent 3c
| S. No. | Solvent | Catalyst (10 mol%) | Yield % |
| 1 | H2O | Fe(acac)3 | 40 |
| 2 | MeOH | Fe(acac)3 | 87 |
| 3 | EtOH | Fe(acac)3 | 95 |
| 4 | DCM | Fe(acac)3 | 75 |
| 5 | AcCN | Fe(acac)3 | 78 |
| 6 | CHCl3 | Fe(acac)3 | 64 |
| 7 | Dioxane | Fe(acac)3 | 58 |
| 8 | EtOH | No catalyst | 20 |
| aReaction conditions: Phenacyl bromide (0.001 mol), o-pheneylenediamine(0.001 mol), 10 mol % Fe(acac)3in 10 ml EtOH, at 60 °C for 50 min. bIsolated yields | |||
The influence of two key experimental parameters, catalystand solvent, was systematically studied. Additionally, the reaction scope was explored by varying phenacyl bromide and and o-pheneylenediaminesubstrates, as illustrated in Scheme 2 and summarized in table 3. The synthesis of quinoxalines was carried out by reacting substituted phenacyl bromides/benzil (2a–2i) (0.001 mol) and o-pheneylenediamine(1a–1b) (0.001 mol) in the presence of Fe(acac)3 as a catalyst. Further, the reaction mass was stirred at 60 ◦C for 40–80 min. afforded quinoxalines (3a–3k) with excellent yields and high purity.
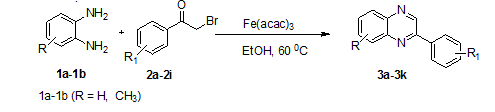
Scheme 2: Synthesis of quinoxalines (3a–3k) using Fe(acac)3 catalyst
Table 2: Screening of reaction condition with respect to substrate
| S. No. | Amine | Phenacyl bromide | Product | Yieldb |
| 1 | o-pheneylene diamine | 4-Chloro Phenacylbromide |
2-Phenylquinoxaline |
88 |
| 2 | o-pheneylene diamine | 4-Fluro phenacylbromide |
2-(4-Fluorophenyl)Quinoxaline |
90 |
| 3 | o-pheneylene diamine | 4-Chloro Phenacylbromide |
2-(4-Chloro phenyl)-Quinoxaline |
94 |
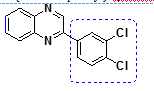
| 4 | o-pheneylene diamine | 3,4 dichloroPhenacylbromide |
2-(3,4-dichlorophenyl)quinoxaline |
93 |
| 5 | o-pheneylene diamine | 4-bromo Phenacylbromide |
2-(4-Bromo phenyl)-Quinoxaline |
92 |
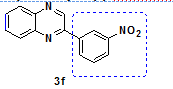
| 6 | o-pheneylene diamine | 3-nitro Phenacylbromide |
2-(3-nitro phenyl)-Quinoxaline |
89 |
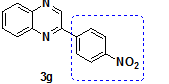
| 7 | o-pheneylene diamine | 4-Nitro Phenacylbromide |
2-(4-Nitro phenyl)-Quinoxaline |
88 |
| 8 | o-pheneylene diamine | 4-Cyano Phenacylbromide |
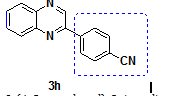
2-(4-Cyano phenyl)-Quinoxaline |
92 |
| 9 | o-pheneylene diamine | 4-Methoxy Phenacylbromide |
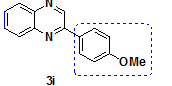
2-(4-mehoxy phenyl)-Quinoxaline |
89 |
| 10 | o-pheneylene diamine | 4-methyl Phenacylbromide |
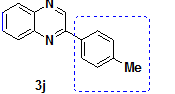
2-(4-Methyl phenyl)-Quinoxaline |
90 |
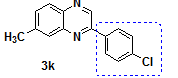
| 11 | 4-methyl-o-pheneylenediamine | 4-Chloro Phenacylbromide |
2-(4-Chloro-phenyl)-7-methyl-quinoxaline |
91 |
| aReaction conditions: Phenacyl bromide (0.001 mol), o-phenylenediamine (0.001 mol), 10 mol % Fe(acac)3 in 10 ml EtOH, at 60 °C for 50 min. bIsolated yields (3a-3k) | ||||
It was observed that both electron-donating and electron-withdrawing substituents on phenacyl bromide substrates facilitated smooth reactions, yielding the desired products (3a-3k) in high yields (88–94%). However, phenacylbromide derivatives containing electron-withdrawing groups such as nitro, fluoro, chloro, bromo, cyano, and exhibited a faster reaction rate compared to those with electron-donating groups such as methyl and methoxy. This suggests that electron-withdrawing substituents enhance the reactivity of phenyl diamine in this transformation. All synthesized products were thoroughly characterized using (IR) spectroscopy, (¹H NMR), (¹³C NMR), and mass spectrometry, for confirming their structures. All data can agreement with reported data Lit [1, 4, 8, 21]
Plausible mechanism for synthesis of quinoxaline by Fe(acac)3 catalyst
The plausible mechanism for the quinoxalines synthesis was Proposes in scheme 3, which involves the protonation of the carbonyl group of phenacyl bromide over Fe(acac)3 catalyst (A). Later on, it reacts with o-pheneylenediaminethat involves dehydration and dehalogenation simultaneously, resulting in the formation of cyclic product B, which is readily oxidized in air to form desired product C [20].
Scheme 3: Plausible mechanism for the synthesis of quinoxaline derivatives
CONCLUSION
In conclusion, we have developed a mild, efficient, and environmentally benign synthetic protocol for the synthesis of quinoxalines (3a–3k) from substituted phenacyl bromides and o-pheneylenediamines using Fe(acac)₃ as a catalyst. A key feature of this protocol is its simple reaction conditions and the absence of side reactions, leading to the formation of the desired products in high yields. This method serves as an effective alternative to conventional processes for the synthesis of quinoxalines with good to excellent yields (88-94%).
ACKNOWLEDGMENT
The authors are grateful to the Principal of Sant Dnyaneshwar Mahavidyalaya, Soegaon, Chhatrapati Sambhajinagar, for providing laboratory facilities, and to the Director of Punjab University, Chandigarh, for providing spectral data.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
The authors are equally contributed to the design, optimization, referencing analysis, interpretation, drafting, and final approval of the manuscript.
Design and Experimental work Suraj Ade, Analysis of Data Sunil Choudhare and Santosh Padghan and Manojkumar Chopadedrafting and Allreaming things as corresponding Author
CONFLICT OF INTERESTS
Declared none
REFERENCES
Corona P, Carta A, Loriga M, Vitale G, Paglietti G. Synthesis and in vitro antitumor activity of new quinoxaline derivatives. Eur J Med Chem. 2009;44(4):1579-91. doi: 10.1016/j.ejmech.2008.07.025, PMID 18774202.
Dietrich B, Diederichsen U. Synthesis of cyclopeptidic analogues of triostin a with quinoxalines or nucleobases as chromophores. Eur J Org Chem. 2005;2005(1):147-53. doi: 10.1002/ejoc.200400548.
Nageswar YV, Reddy KH, Ramesh K, Murthy SN. Recent developments in the synthesis of quinoxaline derivatives by green synthetic approaches. Org Prep Proced Int. 2013;45(1):1-27. doi: 10.1080/00304948.2013.743419.
Pereira JA, Pessoa AM, Cordeiro MN, Fernandes R, Prudencio C, Noronha JP. Quinoxaline, its derivatives and applications: a state-of-the-art review. Eur J Med Chem. 2015;97:664-72. doi: 10.1016/j.ejmech.2014.06.058, PMID 25011559.
Seitz LE, Suling WJ, Reynolds RC. Synthesis and antimycobacterial activity of pyrazine and quinoxaline derivatives. J Med Chem. 2002;45(25):5604-6. doi: 10.1021/jm020310n, PMID 12459027.
Cheng G, Sa W, Cao C, Guo L, Hao H, Liu Z. Quinoxaline 1,4-di-N-Oxides: biological activities and mechanisms of actions. Front Pharmacol. 2016;7:64. doi: 10.3389/fphar.2016.00064, PMID 27047380.
Loriga M, Piras S, Sanna P, Paglietti G. Quinoxaline chemistry part 7.2-[aminobenzoates] and 2-[aminobenzoylglutamate]-quinoxalines as classical antifolate agents synthesis and evaluation of in vitro anticancer, anti-HIV and antifungal activity. Farmaco. 1997;52(3):157-66. PMID 9212450.
Hui X, Desrivot J, Bories C, Loiseau PM, Franck X, Hocquemiller R. Synthesis and antiprotozoal activity of some new synthetic substituted quinoxalines. Bioorg Med Chem Lett. 2006;16(4):815-20. doi: 10.1016/j.bmcl.2005.11.025, PMID 16309903.
Maikhuri VK, Prasad AK, Jha A, Srivastava S. Recent advances in the transition metal catalyzed synthesis of quinoxalines: a review. New J Chem. 2021;45(30):13214-46. doi: 10.1039/D1NJ01442K.
More SV, Sastry MN, Yao CF. Cerium (IV) ammonium nitrate (CAN) as a catalyst in tap water: a simple proficient and green approach for the synthesis of quinoxalines. Green Chem. 2006;8(1):91-5. doi: 10.1039/B510677J.
Bhunia S, Ghosh P, Patra SR. Gold-catalyzed oxidative alkyne functionalization by N−O/S−O/C−O bond oxidants. Adv Synth Catal. 2020;362(18):3664-708. doi: 10.1002/adsc.202000274.
Hasaninejad A, Zare A, Mohammadizadeh MR, Karami Z. Synthesis of quinoxaline derivatives via condensation of Aryl-1,2-Diamines with 1,2-diketones using (NH4)6Mo7O24.4H2O as an efficient, mild and reusable catalyst. JICS. 2009;6(1):153-8. doi: 10.1007/BF03246514.
Danoun K, Essamlali Y, Amadine O, Mahi H, Zahouily M. Eco-friendly approach to access of quinoxaline derivatives using nanostructured pyrophosphate Na2PdP2O7 as a new, efficient and reusable heterogeneous catalyst. BMC Chem. 2020;14(1):6. doi: 10.1186/s13065-020-0662-z.
Pan F, Chen TM, Cao JJ, Zou JP, Zhang W, P. Ga(ClO4)3-catalyzed synthesis of quinoxalines by cycloaddition of α-hydroxyketones and o-phenylenediamines. Tetrahedron Lett. 2012;53(20):2508-10. doi: 10.1016/j.tetlet.2012.02.113.
Mohammadi H, Shaterian HR, R. (3-Oxo-[1,2,4]triazolidin-1-yl)bis (butane-1-sulfonic acid) functionalized magnetic γ-Fe2O3 nanoparticles: a novel and heterogeneous nanocatalyst for one-pot and efficient four-component synthesis of novel spiro[indeno[1,2- b]quinoxaline derivatives. Applied Organom Chemis. 2019;33(6):e4901. doi: 10.1002/aoc.4901.
Gopalaiah K, Saini A, Chandrudu SN, Rao DC, Yadav H, Kumar B. Copper-catalyzed aerobic oxidative coupling of o-phenylenediamines with 2-aryl/heteroarylethylamines: direct access to construct quinoxalines. Org Biomol Chem. 2017;15(10):2259-68. doi: 10.1039/C7OB00122C, PMID 28240337.
Gao J, Ren ZG, Lang JP. One-pot aqueous-phase synthesis of quinoxalines through oxidative cyclization of deoxybenzoins with 1,2-Phenylenediamines catalyzed by a Zwtterionic Cu(II)/calix[4]arene complex. Chin Chem Lett. 2017;28(5):1087-92. doi: 10.1016/j.cclet.2016.12.035.
Bera A, Sk M, Singh K, Banerjee D. Nickel-catalysed dehydrogenative coupling of aromatic diamines with alcohols: selective synthesis of substituted benzimidazoles and quinoxalines. Chem Commun (Camb). 2019;55(42):5958-61. doi: 10.1039/C9CC02319D, PMID 31050346.
Hamid O, Chari MA, Van Nguyen C, Chen JE, Alshehri SM, Yanmaz E. ZnO-loaded mesoporous silica (KIT-6) as an efficient solid catalyst for production of various substituted quinoxalines. Catal Commun. 2017;90:111-5. doi: 10.1016/j.catcom.2016.10.026.
Zhao Z, Yu X, Zhu L, Tan S, Fu W, Wang L. Synthesis of α,β-unsaturated ketones with secondary alcohols and aldehydes catalyzed by Fe(acac)3. Chemistry Select. 2022;7(34):e202202093. doi: 10.1002/slct.202202093.
Zhang R, Qin Y, Zhang L, Luo S. Oxidative synthesis of benzimidazoles, quinoxalines and benzoxazoles from primary amines by Ortho-quinone catalysis. Org Lett. 2017;19(20):5629-32. doi: 10.1021/acs.orglett.7b02786, PMID 28968131.