
Int J Chem Res, Vol 7, Issue 3, 1-4Research Article
PHYTOCHEMICAL CONSTITUENTS AND ANTIOXIDANT ACTIVITY OF PERSEA AMERICANA LEAVE
AMUDA MUTIU OLASUNKANMI1*, ODERINLO OGUNYEMI2
1Department of Pure and Applied Chemistry, Ladoke Akintola University of Technology, Ogbomoso, Oyo State, Nigeria. 2Department of Chemistry, Faculty of Science, Federal University Otuoke, Bayelsa, Nigeria
*Corresponding author: Amuda Mutiu Olasunkanmi; Email: [email protected]
Received: 10 Apr 2023 Revised and Accepted: 25 May 2023
ABSTRACT
Objective: Persea americana is a popular plant that is well known for its fruit, which is highly consumed due to its nutritional and medicinal values. The rich secondary metabolites content of P. americana creates an opportunity to utilize it as a functional food or standardized herbal preparations. The quality of a medicinal plant is determined by the active substances produced by the plant. Therefore, the purpose of the present study is to determine the secondary metabolites present in P. americana grown in uncultivated farmland in Ogbomoso, Oyo State Nigeria.
Methods: The dried pulvurised leaves of P. americana from uncultivated farmland in Ogbomoso, Oyo State Nigeria were extracted with methanol by the maceration method and partitioned successively with solvents of increasing polarity. The extracts obtained were subjected to phytochemical screening and the total phenolics were determined spectrometrically using the folin–ciocalteu method.
Results: The phytochemical screening showed that P. americana is rich in various secondary metabolites such as flavonoids, saponins, tannins, and alkaloids. The total phenolic content gave 500µg/g gallic acid equivalent. The total flavonoid content obtained was 250µg/g using quercetin as standard. The anti-oxidant assay of the crude extract showed a high activity of 89.05% at a concentration of 0.05g/ml.
Conclusion: Total phenolic content of the plant is likely to be a key for determining the free radical scavenging and ROS reducing ability of the plant.
Keywords: Antioxidant, Flavonoid, Phytochemical, Phenolic, Extraction
© 2023 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (http://creativecommons.org/licenses/by/4.0/)
DOI: http://dx.doi.org/10.22159/ijcr.2023v7i3.219. Journal homepage: https://ijcr.info/index.php/journal
INTRODUCTION
Nature is a treasure trove of many bioactive secondary metabolites that are used in the treatment and prevention of a variety of human ailments. These secondary metabolites are also an indispensable source of inspiration in the discovery and development of novel therapeutic agents [1]. The plant kingdom has continually offered a plethora of these natural medicines and it is estimated that about 25% of our current drug arsenals is from higher plants [2, 3]. The traditional system of medicine, which principally centres on herbal medicines, plays an important role in the physical and psychological well-being for the vast majority of the world’s population [4, 5]. Herbal medicine is widely accepted because of its ease of accessibility, comparative cheaper cost and relative safety profiles when compared to the synthetic alternatives [6]. For example, over 80% of the African population relies on herbal medicine [7] either solely or in combination with conventional medicines [1], and the use of herbs is increasingly gaining recognition in western countries [8].
The therapeutic effects of herbs may be attributed to their antioxidant property [9]. Natural antioxidants, such as flavonoids, coumarins, tocopherols, carotenoids, and phenolic acids, are widely distributed in medicinal plants. They exhibit a wide variety of biological effects such as anti-inflammatory, anticancer, anti-aging, antidiabetes e. t. c [10]. Although plant's antioxidant primarily function as free radical scavengers, they also interact with pro-oxidant metals and act as reducing agents [11].
Persea americana, commonly called avocado, is a flowering plant of Lauraceae family and a native to central Mexico. It is well known throughout the world for its fruit which is highly consumed because of its peculiar nutritional composition, antioxidant content, and biochemical profile [12]. Compound class such as carotenoids, tocopherols, phenolics (i.e., flavonoids, condensed tannins and hydroxycinnamic acids), acetogenins monounsaturated and polyunsaturated fatty acids are the main antioxidants found in avocado. Several studies have reported notable positive correlations between the phenolic contents and the antioxidant capacity of avocado [13]. Because it is well known that environmental conditions can influence the phytochemical composition and the quantity of phenolic compounds present in plants [14, 15], the current study examines the phytochemical and antioxidant profiles of P. americana grown in uncultivated farmland in Ogbomoso, Oyo State, Nigeria.
MATERIALS AND METHODS
Reagents and materials
All chemical reagents used where of analytical grade purchased from Sigma Chemical Co. London. Absorbance was measured using AJI-CO3 UV-VIS spectrophotometer.
Sample collection and identification
Fresh leaves of P. americana were collected from uncultivated farmland in Aroje, Ogbomosho in Oyo State (Latitude 8° 07’ 60.00”N, Longitude 4° 14’ 60.00” E). The leaves were taken to a botanist in the Department of Pure and Applied Biology, Ladoke Akintola University of Technology, Ogbomoso, for proper identification and classification (Voucher No. LHO 656). The leaves were then air-dried at room temperature for three weeks.
Extraction of plant material
The dried leaves were pulverized using an electric grinder and 1.7 kg of the pulverized material was extracted by maceration with methanol (1.5 liters). The solvent was filtered off after 24 h and removed in vacuo to give the crude methanol extract. Thereafter, by adapting the approach of Emran and co-workers [16], the crude methanol extract was successively partitioned with increasing polarity solvents in the following order; n-hexane, chloroform, ethyl-acetate, methanol.
Phytochemical screening
The qualitative phytochemical analysis of the extracts from the leaves of P. americana adopted the methods [17, 18] presented as follows:
Test for flavonoids
1 ml of 10% NaOH was added to 1 ml of the extract. A yellow colouration indicates the presence of flavonoids.
Test for steroids (Salkovoski test)
5 drops of concentrated sulphuric acid (H2SO4) was added to 1 ml of the crude extract. Red coloration indicates the presence of steroids.
Test for alkaloids
5 ml of 1% HCl solution was added to 2 mls of the extract in a test tube; the mixture was heated and filtered. The filtrate was used for the following tests;
To 1 ml of the filtrate, 2 drops of Mayer’s reagent was added. A creamy precipitate indicates the presence of alkaloids in the extract.
To 1 ml of the filtrate, 2 drops of Wagner’s reagent was added, a reddish-brown precipitate indicates the presence of alkaloids.
Test for saponins (frothing test)
2 ml of the extract in a test tube was vigorously shaken for about 2 min to observe if there would be frothing. Frothing indicates the presence of saponins.
Test for tannins
1 ml of freshly prepared 10% KOH was added to the plant extract. A dirty white precipitate indicates the presence of tannins in the extract. Alternatively, 2 drops of 5% FeCl3 was added to 1 ml of the extract. A greenish precipitate indicates the presence of tannins.
Test for glycosides
To 1 ml of the extract in a test tube, 10 ml of 50% H2SO4 was added. The mixture was heated in boiling water for 15 min, 10 ml of Fehling’s solutions (I and II) was added and the mixture was boiled. A brick-red precipitate indicates the presence of glycosides in the extract.
Determination of total flavonoid contents
The AlCl3 method (18) was used for determination of the total flavonoid contents of the sample. The extract (1.5 ml) was added to 10 ml volumetric flask filled with 5 ml distilled water and 0.3 (ml) of 5% NaNO2 and mixed. A reagent blank was using distilled water. After 5 min, 1.5 ml of 2% methanolic AlCl3 was added. 2 ml of 1 mol dm-3 NaOH was added 5 min later and then the volume was made up to 10 ml with distilled water. The mixture was shaken for 5 min and after 10 min the absorbance was taken at 420 nm. Flavonoids contents were calculated using a standard calibration curve, prepared from quercetin. The flavonoids contents were expressed as mg quercetin g-1 of extract.
Determination of total phenolic contents
Total phenolic were determine using Folin-Ciocalteu method [18]. The methanolic extracts (0.5 ml) were added to a 25 ml volumetric flask filled with 10 ml distilled water and 2.5 ml of 0.2 Folin-ciocalteu phenol reagents. A reagent blank using distilled water instead of sample was prepared. After 5 min, 2 ml of 2% Na2CO3 solution were added with mixing. The solution was diluted to the volume (25 ml) with distilled water and then allowed to stand for 90 min, and the absorbance was measured at 725 nm. Total phenolic contents were calculated as mg Gallic acid g-1 dry weight of sample.
Anti-oxidant assay
1 ml of various concentrations of the extract in methanol was added to 4 ml of 0.1 mmol methanolic solution of 1, 1-Diphenyl-2-picrylhydrazyl (DPPH). A blank probe was obtained by mixing 4 ml of 0.1 mmol methanolic solution of DPPH and 200 µl of distilled water. After 30 min of incubation in the dark at room temperature, the absorbance was taken at 515 nm against the prepared blank. Inhibition of the free radicals by DPPH in percent (I %) was calculated using this formula.
DPPH in 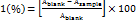
Ascorbic acid was used as standard control. IC50 (inhibition concentration at 50) denotes the concentration of sample which is required to scavenge 50% of DPPH free radicals
RESULTS AND DISCUSSION
Table 1: Phytochemical screening of P. americana leaves extracts
| Extract/Fractions | Steroid | Flavonoid | Saponin | Tannin | Alkaloid | Glycoside |
| n-Hexane | − | − | + | − | + | − |
| Chloroform | − | + | + | − | + | − |
| Ethyl acetate | − | + | + | + | + | + |
| Methanol | + | + | + | − | − | + |
Note: += present; − = absent
Table 2: Total phenolic and flavonoid contents of methanolic extract of P. americana
| Extract | Phenolic content (µg/g) | Flavonoid content (µg/g) |
| Methanolic extract | 500 | 250 |
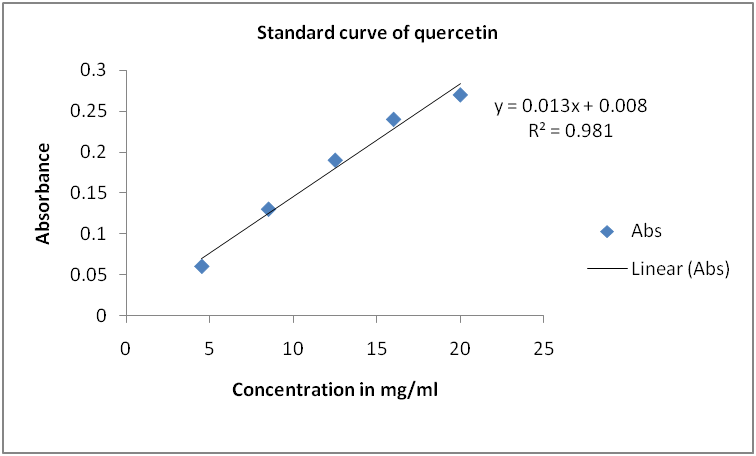
Fig. 1: Standard curve for quercetin
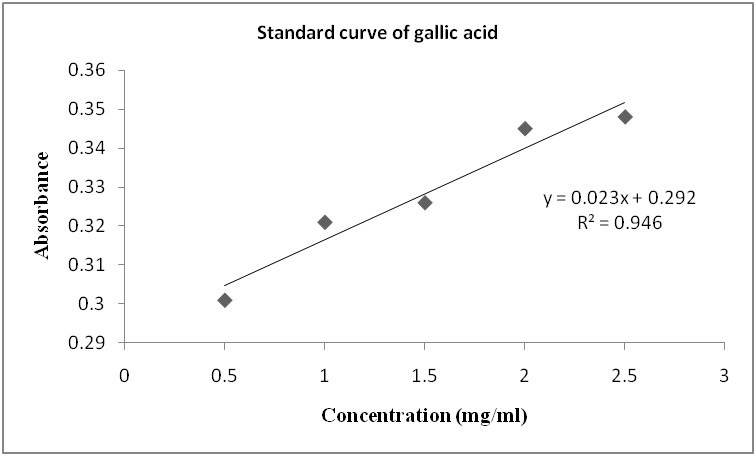
Fig. 2: Standard curve for gallic acid
Table 3: Antioxidant analysis
| S. No. | Absorbance | Concentration | % Inhibition/IC50 |
| 1 | 0.224 | 0.15 | 81.18/6.21 |
| 2 | 0.30 | 0.05 | 89.08/3.36 |
| 3 | 0.320 | 0.20 | 73.11/8.67 |
| 4 | 0.54 | 0.10 | 87.06/7.35 |
DISCUSSION
The phytochemical screening of the extracts (table 1) revealed the presence of flavonoids, saponins, alkaloids, tannins, glycosides, steroids in the leaves extracts of P. americana. This result is in agreement with the previous analysis of P. americana leaves extracts obtained from Akure, Ondo State, Nigeria by Ajayi et al., [19], except for the absence of steroids in their extracts, which is present in our methanol fraction. Similarly, our findings correspond with the analysis of avocado leaf extract from Indonesia by Rahman et al., [12] with the exception that alkaloid is lacking in their extract.
Furthermore, only saponins and alkaloids were found present in our n-hexane extract, whereas flavonoids, tannins, glycosides and steroids were all absent. From the chloroform, ethyl acetate, and methanol fractions, the type of flavonoid present were analysed to be flavones and flavonols.
The present study revealed (table 2) the phenolic content of the leave of P. americana in terms of mg gallic acid equivalent/g dry weight of the sample (standard plot: y= 0.023x+0.292, R2=0.946), the value was 500µg/g gallic acid equivalent/g. Plant phenolics are diverse group of secondary metabolites and are utilized as wound and infection protectants. Phenolics have been identified to have several biological activities such as antioxidants, antimutagenic, anticancinogenic, anti-inflammatory and antimicrobial activities in human [20, 21].
Using the standard curve of quercetin (y= 0.013x+0.008, R2= 0.981), the flavonoid contents of the leaf of P. americana was found to be 250 µg/g quercetin equivalent/g of dry sample weight.
Table 3, indicate free radical scavenging potency of the methanolic extract, which was carried out using a UV-spectrometer at an absorbance of 470 nm with different concentration ranging from 0.20-0.5g. The results shows that the extract is more potent at a lower concentration (0.05g) with %I of (89.08%) up to the higher concentration (0.20g) with %I (73.11%), which is less potent in that trend.
CONCLUSION
The results obtained indicate that P. americana leaves extracts contain a good amount of phenolic and flavonoid content and can be used as a natural source antioxidant that could have great importance as therapeutic agents in preventing or slowing the progress of aging and age-associated oxidative stress. Additionally, phytochemicals can have complementary and overlapping mechanism of action in the body, including anti-oxidant effects, modulation of detoxification enzymes, stimulation of the immune system and modulation of hormone metabolism.
ACKNOWLEDGEMENT
The authors appreciate Ishola Morufu of Davita Inc (2669 Church St, East Point, GA 30344) for his massive support during the course of this research.
FUNDING
Nil
AUTHORS CONTRIBUTIONS
AMO conceptualised the study, collected the plant material, coordinated and performed the experimental. OOO provided intellectual leadership. Both authors participated in the preparation of the manuscript and reviewed the final version.
CONFLICT OF INTERESTS
Declared none
REFERENCES
-
Yuan H, Ma Q, Ye L, Piao G. The traditional medicine and modern medicine from natural products. Molecules. 2016;21(5). doi: 10.3390/molecules21050559, PMID 27136524.
-
Mishra PK, Singh AP, Singh AP. Efficacy of aqueous and methanolic extracts of plant Brassica rapa leaves potential anti-pseudomonal activity. Int J Curr Pharm Sci. 2022/15;14(1):77-82. doi: 10.22159/ijcpr.2022v14i1.44115.
-
Calixto JB. The role of natural products in modern drug discovery. An Acad Bras Cienc. 2019;91 Suppl 3:e20190105. doi: 10.1590/0001-3765201920190105, PMID 31166478.
-
Dubey S, Tiwari P, Samanta K. Preliminary physical-phytochemical and phytocognostical evaluation of the leaves parts and evaluation of herbal ointment using leaves of Moringa oleifera lam leaf extract. Int J Curr Res. 2023 Feb;15(02):23898-903.
-
Basha SK, Sudarsanam G. Traditional use of plants against snakebite in sugali tribes of yerramalais of Kurnool District, Andhra Pradesh, India. Asian Pac J Trop Biomed. 2012;2(2):S575-9. doi: 10.1016/S2221-1691(12)60276-7.
-
Iwu MW, Duncan AR, Okunji CO. New antimicrobials of plant origin. In: Janick J, editor. Perspectives on new crops and new uses. Alexandria: ASHS Press; 1999. p. 457-62.
-
Shewamene Z, Dune T, Smith CA. Use of traditional and complementary medicine for maternal health and wellbeing by African migrant women in Australia: a mixed method study. BMC Complement Med Ther. 2020;20(1):60. doi: 10.1186/s12906-020-2852-6, PMID 32070348.
-
Pan SY, Litscher G, Gao SH, Zhou SF, Yu ZL, Chen HQ. Historical perspective of traditional indigenous medical practices: the current renaissance and conservation of herbal resources. Evid Based Complement Alternat Med. 2014;2014:525340. doi: 10.1155/2014/525340, PMID 24872833.
-
Adesokan AA, Yakubu M, Owoyele B, Akanji M, Soladoye A, Lawal OK. Effect of administration of aqueous and ethanolic extracts of Enantia chlorantha stem bark on brewer’s yeast-induced pyresis in rats. Afr J Biochem Res. 2008;2:165-9.
-
Xu DP, Li Y, Meng X, Zhou T, Zhou Y, Zheng J. Natural antioxidants in foods and medicinal plants: extraction, assessment and resources. Int J Mol Sci. 2017;18(1). doi: 10.3390/ijms18010096, PMID 28067795.
-
Matkowski A, Tasarz P, Szypuła E. Antioxidant activity of herb extracts from five medicinal plants from Lamiaceae, subfamily Lamioideae. J Med Plants Res. 2008;11/01:2.
-
Rahman N, Utami Dewi NUB. Phytochemical and antioxidant activity of avocado leaf extract (Persea americana Mill.). Asian J Sci Res. 2018;11(3):357-63. doi: 10.3923/ajsr.2018.357.363.
-
Bhuyan DJ, Alsherbiny MA, Perera S, Low M, Basu A, Devi OA. The odyssey of bioactive compounds in avocado (Persea americana) and their health benefits. Antioxidants (Basel). 2019;8(10). doi: 10.3390/antiox8100426, PMID 31554332.
-
Yin X, Chavez Leon MASC, Osae R, Linus LO, Qi LW, Alolga RN. Xylopia aethiopica seeds from two countries in West Africa Exhibit Differences in Their Proteomes, Mineral Content and Bioactive Phytochemical Composition. Molecules. 2019;24(10):1979. doi: 10.3390/molecules24101979, PMID 31126018.
-
Kumar S, Yadav A, Yadav M, Yadav JP. Effect of climate change on phytochemical diversity, total phenolic content and in vitro antioxidant activity of Aloe vera (L.) Burm.f. BMC Res Notes. 2017;10(1):60. doi: 10.1186/s13104-017-2385-3, PMID 28118858.
-
Emran TB, Rahman MA, Uddin MMN, Rahman MM, Uddin MZ, Dash R. Effects of organic extracts and their different fractions of five Bangladeshi plants on in vitro thrombolysis. BMC Complement Altern Med. 2015;15(1):128. doi: 10.1186/s12906-015-0643-2, PMID 25902818.
-
Nciki S, Oderinlo OO, Gulube Z, Osamudiamen PM, Idahosa KC, Patel M. Mezoneuron benthamianum inhibits cell adherence, hyphae formation, and phospholipase production in Candida albicans. Arch Microbiol. 2020;202(9):2533-42. doi: 10.1007/s00203-020-01972-2, PMID 32656677.
-
Jagadish LK, Krishnan VV, Shenbhagaraman R, Kaviyarasan V. Comparative study on the antioxidant, anticancer and antimicrobial property of Agaricus bisporus (J.E. Lange) Imbach before and after boiling. Afr J Biotechnol. 2009;8(4):654-61.
-
Ajayi O, Awala S, Olalekan O, Alabi O. Evaluation of antimicrobial potency and phytochemical screening of Persea americana leaf extracts against selected bacterial and fungal isolates of clinical importance. Microbiol Res J Int. 2017;20(1):1-11. doi: 10.9734/MRJI/2017/24508.
-
Dhalaria R, Verma R, Kumar D, Puri S, Tapwal A, Kumar V. Bioactive compounds of edible fruits with their anti-aging properties: A comprehensive review to prolong human life. Antioxidants (Basel). 2020;9(11). doi: 10.3390/antiox9111123, PMID 33202871.
-
Shahidi F, Ambigaipalan P. Phenolics and polyphenolics in foods, beverages and spices: antioxidant activity and health effects–a review. J Funct Foods. 2015;18:820-97. doi: 10.1016/j.jff.2015.06.018.