
Int J Chem Res, Vol 7, Issue 2, 13-18Research Article
TOTAL PHENOLIC, FLAVONOID AND MINERAL CONTENTS OF THE METHANOLIC LEAF EXTRACT OF PARINARI CURATELLIFOLIA PLANCH. EX BENTH
EDEWOR THERESA IBIBIA1*, AGBOOLA PETER OBALOLUWA1, AMUDA MUTIU OLASUNKANMI1, MMUO AGATHA IJEOMA1, OWA STEPHEN OLUGBEMIGA2
1Department of Pure and Applied Chemistry, Ladoke Akintola University of Technology, Ogbomoso, Oyo State, Nigeria, 2Department of Biological Sciences, Landmark University, Omu aran, Kwara State, Nigeria
Email: [email protected]
Received: 25 Dec 2022 Revised and Accepted: 21 Feb 2023
ABSTRACT
Objective: Parinari curatellifolia is a medicinal plant that possesses multifunctional properties. The aim of this research was to evaluate the mineral, total phenolic and flavonoids contents and some biological parameters of the methanolic leaf extract.
Methods: The leaves were extracted with methanol to obtain the crude extract. Phytochemical screening was carried out using the method described by Harborne, 1999. The extract was subjected to GC-MS analysis, Aluminum colorimetric assay was used to determine the total flavonoid content while Folin-Ciocalteau reagent was used to evaluate the total phenolic content. The antioxidant activity was determined using the DPPH radical scavenging assay. The mineral content was determined via atomic absorption spectrophotometer. Disc diffusion method was used for the antibacterial assay.
Results: The phytochemical screening revealed the presence of tannins, saponins, flavonoids, terpenoids, alkaloids and cardiac glycosides, while steroids, anthraquinones and cyano glycosides were absent. The GC-MS profile showed catechol as the most abundant phytochemical and the identified phytocomponents were mainly polyphenolics, carbonyls, fatty acids and fatty acid esters. The extract possesses total flavonoid and phenolic contents of 13.46±0.02 mg/g quercetin equivalent and 58.43±0.15 mg/g Gallic acid equivalent, respectively. The extract exhibited a significant antioxidant of 84%. Concentration of the macro and micro elements were quite high in some cases. Na had the highest concentration of 42522.70±2146.50 µg/g, while Cu has the lowest value of 4.18±0.03µg/g. The antibacterial activity of the extract was comparable to that of the standard antibiotic. Bacillus subtilis exhibited the highest zone of inhibition of 29±0.10 mm while Salmonella typii and Kliebsiella pneumoniae gave the lowest value of 14±0.12 mm.
Conclusion: Parinari curatellifolia leaves possess tannins, saponins, flavonoids, terpenoids, alkaloids and cardiac glycosides. It has high antioxidant and antibacterial capabilities.
Keywords: Parinari curatellifolia, Phytochemicals, Phenolic, Flavonoid, Antioxidant, Antibacterial, Elements, GC-MS
© 2023 The Authors. Published by Innovare Academic Sciences Pvt Ltd. This is an open access article under the CC BY license (http://creativecommons.org/licenses/by/4.0/)
DOI: http://dx.doi.org/10.22159/ijcr.2023v7i2.218. Journal homepage: https://ijcr.info/index.php/journal
INTRODUCTION
A large number of synthetic antimicrobial agents are available for the treatment of diseases caused by microorganisms. But researchers have shown that most of these microorganisms are showing resistance to the available synthetic antimicrobial agents. These synthetic antimicrobials also have terrible side effects on the recipients. So there is the need to develop save antimicrobial agents that can effectively combat infective microorganisms [1, 2]. Moreover, endogenous metabolic processes in the human body and exposure to chemicals in our environment lead to the generation of reactive oxygen species such as superoxide radical, hydrogen peroxide, hydroxide radicals and peroxy radicals. These reactive species are involved in the pathogenesis of diseases such as cancer, cardiovascular disorder, atherosclerosis, neurodegenerative disorder and inflammation [3]. Studies on plants that possess medicinal properties show that these plants contain phytoconstituents such as flavonoids, alkaloids, tannins, etc which are responsible for the biological and pharmacological activities of these plants. They have also been shown to be valuable in the prevention of disorders, reduction of diseases and maintenance of health. Some of these compounds have the ability to scavenge free radicals or inhibit electron transfer. They donate hydrogen atom to free radicals and chelate metal ions such as Fe3+and Cu2+; which is an important pharmacological activity. They can also up-regulate antioxidant enzymes. The use of medicinal plants in the treatment of ailments is more predominant in underdeveloped countries than in developed countries. This is due to the fact that synthetic drugs are easier and cheaper to obtain in developed countries and more traditional health practitioners’ are found in under-developed countries where the synthetic drugs are out of reach of the common man.
Parinari curatellifolia is a medicinal plant that grows well in Africa (west, east, central and south). It is an evergreen tree that stands up to 25 m. The leaves are simple, alternate, elliptical to oblong and leathery. The top of the leaves are darker in green color than the underneath. The apex tapers broadly, base is square, margin entire and the petiole short. The leaves have been reported to possess sedative, antidiabetic properties. Decoction of the leaves is drunk for the treatment of fever, while the crushed leaves are used for dressing of fractures, dislocations, wounds, cuts and sores [4]. The leaf extract could also be used as an expectorant, antibacterial and anti-inflammatory [5]. Other properties include its use for dental hygiene [6], constipation [7] and hypertension [8]. The objective of this research is to carry out both chemical and biological analysis of the leaves of Parinari curatellifolia in order to determine the phytochemicals, minerals and some biological parameters of the methanolic leaf extract.
MATERIALS AND METHODS
General
The following chemicals methanol, sodium hydroxide, sodium carbonate, aluminum oxide, sodium nitrite, Folin-ciocalteau reagent, potassium acetate, were obtained from E. Merck (Darmstadt, Germany) while quercetin, Gallic acid, ascorbic acid and 2, 2-diphenyl-1-picrylhydrazyl (DPPH) were obtained from Sigma Chemical Co. (St. Louis, MO, USA). Absorbance measurements were recorded on a Genseys 10S vI. 200 217H311008 spectrophotometer.
Sample collection and preparation
The plant leaves were collected from an herbalist in Omu aran, Kwara State, Nigeria. Omu aran is located on longitude 5.0963 °E and latitude 8.1402 °N. It was identified by matching the local name of the plant with that found in the book “Vernacular names of Nigerian Plants Yoruba” written by Gbile and Soladoye, 2012 [9]. It was later taken to the department of Pure and applied Biology, Ladoke Akintola University of Technology, Ogbomoso, Nigeria for further identification by a plant taxonomist, Prof. A. T. J. Ogunkunle. A voucher specimen was deposited in their herbarium and specimen number of LHO 605 attached to it. The leaves were stripped off the branches, taken to the laboratory and air-dried for two weeks. The dried leaves were pulverized into a fine powder using a Mouliner food blender and stored in an airtight container until further use.
Determination of elements present in the leaves
A mixture of HNO3 and H2O2 in the ratio of 1:1 was introduced into beakers containing 5g of the pulverized leaves. The beakers were placed on hot plates in a fume cupboard and heated until the content turned colorless. The colorless liquid was transferred into a 50 ml volumetric flask; de-ionized water added and made up to the mark. Aliquots of this solution were analyzed for the presence of metals using atomic absorption spectrophotometer Parkin Elmer model 403, USA.
Extraction
500 g of the powdered leaves of Parinari curatellifolia was extracted with methanol using a Soxhlet extractor. The extract was concentrated by using a rotary evaporator. The extract obtained was weighed, stored in a sample bottle and placed in a cool dry place.
Phytochemical screening
The methanol extract was screened for the presence of steroids, flavonoids, tannins, saponins, glycosides, terpenoids and alkaloids according to the method described by Harborne, 1999 [10].
Determination of total flavonoid content
10 ml of methanol was added to 10 mg of the extract and mixed thoroughly using a vortex mixer. 0.5 ml of this solution was pipetted into a test tube and 1.5 ml methanol, 0.1 ml of 10% AlCl3, 0.1 ml potassium acetate and 2.8 ml of distilled water were added to it. The mixture was stirred properly and incubated for 30 min at room temperature. The absorbance was measured at 415 nm against a reagent blank. The total flavonoid content was calculated from the calibration curve of quercetin and expressed as mg of quercetin per gram weight of the dry extract.
Determination of total phenolic content
5 mg of the sample was dissolved in 5 ml of methanol using a vortex mixer. 0.5 ml of this solution was pipetted into a test tube and 3.5 ml of distilled water, 0.25 ml Folin-Ciocalteau reagent added to it. The mixture was incubated for 10 min at room temperature. Later, 0.75 ml of 20% Na2CO3 was added to it and left to incubate for 2h. The absorbance was measured at 765 nm against a reagent blank. The total phenolic content was calculated from the calibration curve of Gallic acid and expressed as mg of Gallic acid per gram weight of dry extract.
GC-MS analysis
A gas chromatography (7890A) from Agilent USA hyphenated to a mass spectrophotometer (5975C) with a triple-axis detector equipped with an auto injector (10 µl syringe) was used. Helium gas was used as the carrier gas. The chromatographic separation was performed on a capillary column having the following specifications: length: 30 m; internal diameter: 0.2 µm; thickness: 250 µm, treated with phenyl methyl siloxane. Other GC-MS conditions are ion source temperature (EI): 250 °C; interface temperature: 300 °C; pressure: 16.2 psia; out time: 1.8 min. 1 µl injector in split mode with split ratio of 1:5 and injection temperature of 300 °C were employed. The column temperature was started at 35 °C for 5 min and changed to 150 °C at the rate of 4 °C. The temperature was later raised to 250 °C at the rate of 20 °C/min and held for 5 min. The total elution time was 47.5 min. MS solution software provided by the supplier was used to control the system and acquire data. The identification of the separated compounds was carried out by comparing the mass spectra obtained with those of the standard mass spectra from NIST library.
Antibacterial analysis
Microorganisms used
The microorganisms used were collected from Bowen teaching hospital, Ogbomoso, Nigeria. These include Escherichia coli, Salmonella typhi, pseudomonas aeruginosa, Bacillus subtilis, Kliebsella pneumoniae, Bacillus aureus and Staphylococcus aureus.
Test samples
1 mg of the methanol extract and the standard antibiotic were separately dissolved in I ml of distilled dimethylsulphoxide.
The antibacterial medium
100 ml of distilled water was added to 2.8 g of nutrient agar, stirred for 5 min and placed in an autoclave at a temperature of 121 °C for 15 min to sterilize. It was later cooled to 45 °C and introduced into sterile petri dishes to solidify.
Disc diffusion test
The nutrient agar plates were inoculated with the test organism and sterile Whatman No. 1 filter paper which has been impregnated with the methanol extract, was placed on the surface of the plates. The plates were incubated at 37 °C and zones of inhibition of the test organisms were determined after 24 h. The same test was carried out for the standard antibiotic, streptomycin and all results obtained as triplicates.
Antioxidant activity by DPPH radical scavenging assay
10 mg of the crude extract was dissolved in 20 ml of methanol using a vortex mixer to give a sample concentration of 0.5 mg/ml. From here different concentrations of the sample were prepared (50 µg/ml, 100 µg/ml, 150 µg/ml, 200 µg/ml, 250 µg/ml, 300 µg/ml, 350 µg/ml).
DPPH was prepared by dissolving 4 mg of DPPH in 100 ml methanol. The control contains no extract, only methanol and 2 ml of DPPH. To each of the different concentrations of the extract was added 2 ml of the prepared DPPH solution in methanol. These were left in the dark for 20 min for color development. Then the absorbance was measured at 517 nm against a reagent blank. Ascorbic acid was used as the standard.
RESULTS AND DISCUSSION
Introduction of very sensitive analytical tools has aided the increased interest in plant's secondary metabolites. Many of these secondary metabolites have been found to possess both biological and pharmacological activities. In this present investigation of Parinari curatellifolia leaves, the phytochemical components present in the methanolic leaf extract were determined. The leaves were shown to possess tannins, saponins, terpenoids, cardiac glycosides, alkaloids and flavonoids while steroids, anthraquinones and cyanogenic glycosides were absent, as presented in table 1.
Table 1: Phytochemical screening of methanolic leaf extract of Parinari curatellifolia
| Extract | Tan | Ste | Sap | Ter | Alk | Anthr | Cy gly | Ca gly | Fla |
| met | + | - | + | + | + | - | - | + | + |
NOTE: met–methanol; tan–tannins; ste–steroids; sap–saponins; ter–terpenoids; alk–alkaloids; Anthr–anthraquinones; Cy gly–cyano glycosides, Ca gly–cardiac glycosides; fla–flavonoids.
This result is in agreement with those of Halilu et al., 2010 [11] but with the exception of terpenoids which was not identified in that of Peni et al., 2010 [12]. Phytochemicals such as alkaloids have been shown to possess physiological activities [13] while flavonoids and tannins possess antioxidant activity [14-16]. Saponins exhibit permeabilizing of cell membrane, lowering of serum cholesterol levels, and stimulation of luteinizing hormone release leading to abortifacient properties and immunomodulatory potential via cytokine interplay [17, 18]. In the determination of the mineral content of the methanolic leaf extract, the concentrations of the macro and trace elements were quite high as shown in table 2. Ca is important in the development of teeth and healthy bones while Na and K are necessary for the repair of worn-out tissues and the building of red blood cells. Zn and Mn are important in enzyme metabolism. Pd and Cd, which are heavy metals, were also detected but in low concentrations as compared to the daily intake of 10 mg/Kg [19]. Although these concentrations are low, one need to be careful in the administration of the plant leaf extract since concentrations of these heavy metals in lower concentrations have been found to be toxic [10].
Table 2: Mineral content of the leaves of Parinari curatellifolia
| Minerals | Content (µg/g) |
| Na | 42522.70±2146.50 |
| K | 3284.36±434.36 |
| Ca | 7058.37±615.55 |
| Al | 62.51±0.49 |
| Mn | 21.74±0.14 |
| Cr | 35.14±0.43 |
| Pb | 10.78±0.21 |
| Cu | 4.18±0.03 |
| Co | 26.70±0.42 |
| Cd | 6.15±0.40 |
| Ni | 26.98±0.21 |
| Zn | 100.37±0.69 |
Note: Results are mean of triplicate and SD
In the determination of the total flavonoid content, an aluminum colorimetric assay was used with quercetin as the standard flavonoid compound. A standard calibration curve was obtained for quercetin (y = 0.012x–0.011) with the coefficient of determination R2 = 0.992 (fig. 1). The total flavonoid content of the leaf extract obtained is presented in table 3 and expressed in mg quercetin equivalent/g extract. The total phenolic content of the leaf extract was determined from a linear Gallic acid standard curve (y = 0.023x+0.292, R2 = 0.946) (fig. 2) and expressed in mg Gallic acid equivalent/g extract (table 3). In the in vitro determination of the antioxidant potential of the methanolic leaf extract, DPPH assay was used. The extract gave a value of 111.17µg/g of ascorbic acid equivalent, while the percentage inhibition of the free radical was 84%. This activity is a result of the presence of polyphenolics in the extract. The free radical scavenging activity was compared to that of ascorbic acid, which is a standard antioxidant. Oxygen plays a very important role in human metabolism and, once inhaled, it undergoes a gradual reduction process and ultimately gets metabolized into water. In this process of absorption, transport, and metabolism, some reactive oxygen species are formed, which can promote lipid peroxidation of unsaturated fatty acids and damage membrane proteins, leading to decreased membrane permeability, decreased activities of enzymes and receptors, and activation of cells. Furthermore, when free radicals react with DNA, cancer-causing mutations may occur. Therefore, antioxidant defense systems in foods and drugs are important in the prevention of many diseases, such as cancer, atherosclerosis, diabetes, and liver cirrhosis etc.
Table 3: Total flavonoid, phenolic and antioxidant activity of the methanol extract of Parinari curatellifolia
Total flavonoid content (mg/g quercetin equivalent) |
Total phenolic content (mg/g Gallic acid equivalent) |
Antioxidant activity of extract (µg/mg Ascorbic acid equivalent) |
| 13.46±0.02 | 58.43±0.15 | 111.17±0.03 |
Note: Results are mean of triplicate and SD
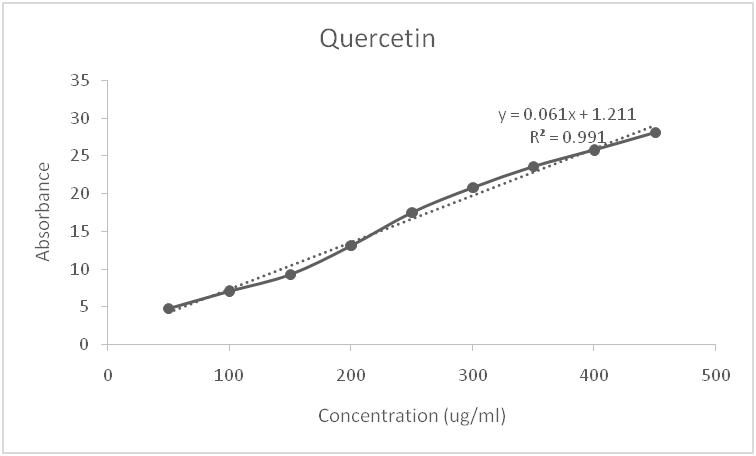
Fig. 1: Standard curve for quercetin
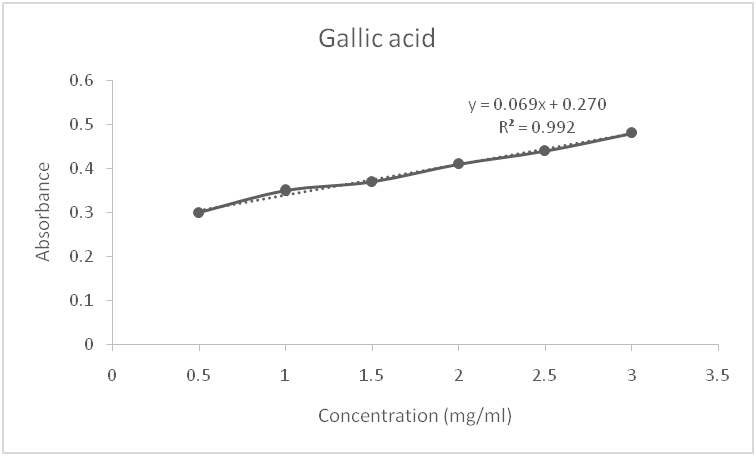
Fig. 2: Standard curve for gallic acid
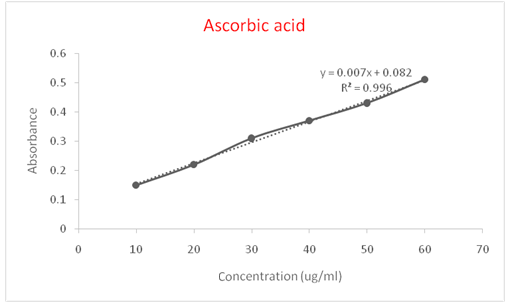
Fig. 3: Standard curve for ascorbic acid
In the last decades GC-MS has been established as a key technological tool for profiling of secondary metabolites in both plants and non-plant species [20-22]. The present study was able to predict, and identify molecular formula and structure of some of the chemicals present in the methanolic leaf extract as presented in table 4. A total of thirty-seven compounds were separated by the GC as shown in the chromatogram in fig. 4. Twenty-five compounds were identified while twelve were unidentified. The MS of the unknown compounds were compared with those of NIST-2011 (National Institute of Standards and Technology) library stored in the computer database of the GC-MS equipment. These compounds were identified based on their molecular weight, peak area in percentage, retention time and molecular formula. The first compound to emerge was 5-methyl-2-furancarboxaldehyde with a retention time 9.53 min while the last to emerge was 2, 2, 4-trimethyl-3-(3, 8, 12, 16-tetramethylheptadeca-3, 7, 11, 15-tetraenyl) cyclohexanol with retention time of 44.75 min. The most abundant compound was catechol (14%) with the retention time of 20.263 min while the least was 2-hydroxy-3-methyl-2-cyclopenten-1-one (0.44%). Other compounds with an appreciable percentage in the methanolic extract are hexadecanoic acid (10.9%), 13-octadecenoic acid (7.9%) and 2-vinylfuran (5.7%). The GC-MS report revealed that the identified compounds are carbonyls, phenolics, fatty acids and fatty acid esters. The presence of these compounds in the plant leaves may be responsible for the plant medicinal properties.
Table 4: GC-MS report of the methanolic leaf extract of Parinari curatellifolia
| S. No. | Name of compounds | Retention time(min) | Peak height | Molar weight | Molecular formula |
| 1 | 5-methyl-2-furancarboxaldehyde | 9.525 | 3714336 | 110 | C6H6O2 |
| 2 | 2-vinylfuran | 11.575 | 7899062 | 94 | C6H6O |
| 3 | Acetic acid, bromophenyl ester | 11.905 | 2029060 | 216 | C8H8O2Br |
| 4 | 2-hydroxy-3-methyl-2-cyclopenten-1-one | 12.824 | 7505568 | 111 | C6H7O2 |
| 5 | 5, 5-dimethyl-2,4-imidazolidione | 13.076 | 7644985 | 128 | C5H8N2O2 |
| 6 | Phenol, 2-methoxy | 14.592 | 10189994 | 124 | C1H8O2 |
| 7 | Maltol | 15.739 | 1731317 | 126 | C6H6O3 |
| 8 | Catechol | 20.263 | 11577713 | 110 | C6H6O2 |
| 9 | 3,4-dihydroxyacetophenone | 21.347 | 2029316 | 152 | C8H8O3 |
| 10 | 2-methoxy-4-vinylphenol | 22.581 | 8704896 | 150 | C9H10O2 |
| 11 | Formic acid, 2, 6-dimethoxyphenyl ester | 23.924 | 7020726 | 182 | C9H10O4 |
| 12 | 2-methoxy-4-(propenyl) phenol | 26.862 | 3058810 | 164 | C10H12O2 |
| 13 | 2, 6-bis(1, 1-dimethylethyl) phenol | 29.014 | 1315212 | 206 | C14H22O |
| 14 | 6-butyl-1,2,3,4-tetrahydronaphthalene | 29.462 | 1573974 | 188 | C14H20 |
| 15 | (z, z)-9,12-octadecadienoic acid | 37.482 | 3672277 | 280 | C18H32O2 |
| 16 | 3,7,11,15-tetramethyl-2-hexadecen-1-ol | 37.710 | 9075171 | 294 | C20H38O |
| 17 | 3,7,11,15-tetramethyl-2-hexadecen-1-ol | 37.906 | 21675194 | 294 | C20H38O |
| 18 | Hexadecanoic acid, methyl ester | 38.370 | 11895225 | 270 | C17H34O2 |
| 19 | n-hexadecanoic acid | 38.904 | 36297509 | 256 | C16H32O2 |
| 20 | Phytol | 39.729 | 8848327 | 296 | C20H40O |
| 21 | Trans-13-octadecenoic acid | 39.980 | 29164227 | 282 | C18H34O2 |
| 22 | Octadecanoic acid | 40.106 | 23769888 | 284 | C18H36O2 |
| 23 | Diisooctylphthalate | 42.132 | 23515947 | 374 | C24H38O3 |
| 24 | 2,2,4-trimethyl-3-(3,8,12,16-tetramethylheptadeca-3,7,11,15-tetraenyl)cyclohexanol | 44.748 | 7974996 | 427 | C30H51O |
Fig.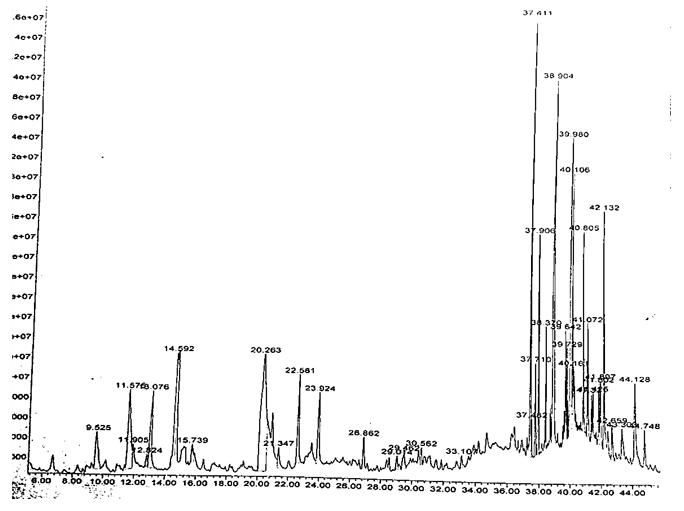 4: Chromatogram of methanolic leaf extract of Parinari curatellifolia
4: Chromatogram of methanolic leaf extract of Parinari curatellifolia
In the study of the antibacterial potential of the methanolic leaf extract, some selected pathogens were screened as shown in table 5. The activity of the methanolic extract was compared with that of a standard antibiotic, streptomycin. The extract exhibited a significant inhibitory effect on the screened bacteria. It gave the highest activity with B. subtilis and the lowest with S. typii and K. pneumoniae. Comparing its activity with that of streptomycin, it exhibited a higher activity with E. coli, K. pneumoniae and B. subtilis but lower activity with S. typii. The activity of S. aureus and B. aureus were comparable to that of streptomycin.
Table 5: Antibacterial activity of the methanolic leaf extract of Parinari curatellifolia
| Bacteria | Zones of inhibition (mm) | |
| Methanol extract | Streptomycin | |
| Escherichia coli | 16±0.10 | 0.00 |
| Staphylococcus aureus | 22±0.14 | 21±0.30 |
| Pseudomonas aeruginosa | 15±0.10 | 19±0.40 |
| Kliebsiella pneumoniae | 14±0.12 | 10±0.20 |
| Bacillus subtilis | 29±0.10 | 25±0.15 |
| Bacillus aureus | 20±0.12 | 22±0.10 |
| Salmonella typii | 14±0.12 | 26±0.20 |
NOTE: Results were mean of triplicate analysis and SD.
CONCLUSION
Our study has shown that the plant leaves of Parinari curatellifolia have a high level of macro and microelements. They are rich in phytochemicals that possess both pharmacological and biological activities. The presence of these phytochemicals is responsible for its high level of antioxidant activity. The GC-MS profile showed catechol as the most abundant phytochemical and the identified phytocomponents were mainly polyphenolics, carbonyls, fatty acids and fatty acid esters. The leaf extract exhibited significant inhibitory activity against the screened microorganisms. Parinari curatellifolia may be considered as a potential source of compounds that may serve as antioxidant, antibacterial and anti-inflammatory agents.
DATA AVAILABILITY
Not applicable
FUNDING
Nil
AUTHORS CONTRIBUTIONS
Edewor T. I designed the research and edited the write-up, Agboola P. O, Amuda M. O and Mmuo A. I carried out the analysis while Owa S. O obtained the plant sample and was responsible for its authentication.
CONFLICT OF INTERESTS
The authors declare that there is none.
REFERENCES
-
Huttner A, Harbarth S, Carlet J, Cosgrove S, Goossens H, Holmes A. Antimicrobial resistance: A global view from the 2013 world healthcare-associated infections forum. Antimicrob Resist Infect Control. 2013;2:31. doi: 10.1186/2047-2994-2-31, PMID 24237856.
-
Roca I, Akova M, Baquero F, Carlet J, Cavaleri M, Coenen S. The global threat of antimicrobial resistance: science for intervention. New Microbes New Infect. 2015;6:22-9. doi: 10.1016/j.nmni.2015.02.007, PMID 26029375.
-
Yao X, Zhang T, Tang H, Wang H, Chen W. Free radical-scavenging, nitrite-scavenging, and N-nitrosoamine formation inhibitory activities of extracts from Kunlun composite tea (Coreopsis tinctoria Nutt.). J Food Nutr Res. 2015;3(9):587-92.
-
Ogbonnia SO, Nkemehule FE, Anyika EN. Evaluation of acute subchronic toxicity of starchytarpheta angustifolia (Mill.) extract in animals. J Biotechnol. 2009;8(9):1793-9.
-
Tochukwu E. Anti-bacterial effect of different fractionations of acetic acid leaf extract of parinari curatellifolia. Br J Pharm Res. 2013;3(2):195-201. doi: 10.9734/BJPR/2013/1772.
-
More G, Tshikalange TE, Lall N, Botha F, Meyer JJ. Antimicrobial activity of Medicinal Plants against oral microorganisms. J Ethnopharmacol. 2008;119(3):473-7. doi: 10.1016/j.jep.2008.07.001, PMID 18672045.
-
Maroyi A. An ethnobotanical survey of medicinal plants used by the people in Nhema communal area, zimbabwe. J Ethnopharmacol. 2011;136(2):347-54. doi: 10.1016/j.jep.2011.05.003, PMID 21575701.
-
Olaleye MT, Amobonye AE, Komolafe K, Akinmoladun AC. Protective effects of Parinari curatellifolia flavonoids against acetaminophen-induced hepatic necrosis in rats. Saudi J Biol Sci. 2014;21(5):486-92. doi: 10.1016/j.sjbs.2014.06.005. PMID 25313285.
-
Gbile ZO, Soladoye MO. Vernacular names of Nigerian plants (Yoruba). 2nd ed. Ibadan: Forestry Research Institute of Nigeria; 2002.
-
Harborne JB. Phytochemical methods. A guide to modern technique in plant analysis. London: Chapman & Hall; 1999. p. 60-6.
-
Halilu ME, Yebpella GG, Hassan LG, Achor M. Preliminary screening, antibacterial and elemental analysis of the leaves and root bark of Parinari curatellifolia planch. ex benth. (Chrysobalanaceae). Continential J Pharm Sci. 2010;4:1-5.
-
Peni J, Elinge CM, Yusuf H, Itodo AU, Agaie BM, Mbongo AN. Phytochemical screening and antibacterial activity of Parinari curatellifolia stem extract. J Med Plants Res. 2010;4(20):2099-102.
-
Doughari JH. Antimicrobial activity of tamarindus indica linn. Trop J Pharm Res. 2006;5(2):597-600. doi: 10.4314/tjpr.v5i2.14637.
-
Zheng W, Wang SY. Antioxidant activity and phenolic compounds in selected herbs. J Agric Food Chem. 2001;49(11):5165-70. doi: 10.1021/jf010697n, PMID 11714298.
-
Kelledar SG, More HN. Estimation of tannins in different parts of Memecyclon umbellantum burm. J Pharm Res. 2010;3(3):554-6.
-
Anandhi D, Srinivasan PT, Kumar GP, Jagatheesh S. Influence of flavonoids and glycosides from Caesalpinia coriariae (Jacq.) wild as antibacterial compound. Int J Curr Microbiol Appl Sci. 2014;3(4):1043-51.
-
Setzer WN, Setzer MC. Plant-derived triterpenoids as potential antineoplastic agents. Mini Rev Med Chem. 2003;3(6):540-56. doi: 10.2174/1389557033487854, PMID 12871157.
-
Heisler I, Sutherland M, Bachran C, Hebestreit P, Schnitger A, Melzig MF. Combined application of saponin and chimeric toxins drastically enhances the targeted cytotoxicity on tumor cells. J Control Release. 2005;106(1-2):123-37. doi: 10.1016/j.jconrel.2005.04.006, PMID 15935506.
-
WHO. Trace elements in human nutrition and health. Technical report series. Geneva; 1996. p. 119-205.
-
Robertson DG. Metabonomics in toxicology: a review. Toxicol Sci. 2005;85(2):809-22. doi: 10.1093/toxsci/kfi102, PMID 15689416.
-
Fernie AR, Trethewey RN, Krotzky AJ, Willmitzer L. Metabolite profiling: from diagnostics to systems biology. Nat Rev Mol Cell Biol. 2004;5(9):763-9. doi: 10.1038/nrm1451, PMID 15340383.
-
Kell DB, Brown M, Davey HM, Dunn WB, Spasic I, Oliver SG. Metabolic footprinting and systems biology: the medium is the message. Nat Rev Microbiol. 2005;3(7):557-65. doi: 10.1038/nrmicro1177, PMID 15953932.